Stories of Hope
Find stories of hope, told by the people who motivate us every day to continue changing the standard in cancer care.



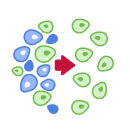
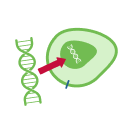
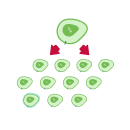
Cell Therapy Explained in 5 Steps
Each cell therapy we develop is uniquely designed for each patient, harnessing the power of the immune system to target and attack cancer. Our CAR T-cell therapy manufacturing process includes these 5 steps:




Newsroom
Recent Press Releases
-
June 01, 2025
Kite Presents New Real-World Data Supporting Use of Potentially Curative Yescarta® in Outpatient Care Setting for Patients with Relapsed/Refractory Large B-Cell Lymphoma at ASCO 2025 -
May 15, 2025
Gilead and Kite Announce Presentation of Transformative Data in 1L Metastatic Triple-Negative Breast Cancer, Updated Results in Multiple Myeloma and Early Data From Novel Investigational CAR T-Cell Therapy Targeting Brain Cancer at 2025 ASCO and EHA -
December 09, 2024
Kite’s Yescarta® Only CAR T-Cell Therapy to Show Durable Response and Long-Term Survival After Five Years in Patients With Relapsed/Refractory Non-Hodgkin Lymphomas at ASH 2024 -
December 09, 2024
Longest Follow-Up Data Reported for Kite’s Tecartus® CAR T-Cell Therapy at ASH 2024 Reinforce Durable Efficacy and Survival Benefits